Panel discussion – Should I move from helium to hydrogen carrier gas?
22 November 2022
The current problems with helium supply are just the latest in a sequence of shortages, which are causing problems for many scientists. But should GC–MS analysts be making the switch to using hydrogen instead? This was the topic of a panel discussion, “Helium shortage 4.0: Is hydrogen carrier gas the solution to the GC–MS problem?” hosted by Separation Science, and held in September 2022. In this blog post I’ll summarise the topics covered, and hope to show that there’s a lot to gain by moving to hydrogen.
The background: why ‘Helium shortage 4.0’?
Well, as the name suggests, this isn’t the first time we’ve experienced issues with helium availability, with the current drop in supply dating back to July 2021, when maintenance work on a helium enrichment unit in the US led to a 10% drop in worldwide helium supply capacity – a problem compounded by breaks in supply from other plants in Russia and Qatar.
However, unlike previous shortages where the main challenge was an increasing cost of helium, ‘Helium shortage 4.0’ has not only meant an increase in price but in many instances a significant reduction in supply, in many cases down by two-thirds. Naturally, this has had a major impact on laboratories that have relied upon helium as a carrier gas for gas chromatography–mass spectrometry (GC–MS) and of course thermal desorption (TD) preconcentration systems.
Concerns over hydrogen carrier gas
When it comes to alternatives to helium as a carrier gas for GC, nitrogen and hydrogen are the obvious candidates. They’re both more readily available than helium and have the distinct advantage that they can easily be produced in the lab using generators. However, the linear velocities for nitrogen are very limited, making it much less efficient than helium or hydrogen when used for TD and/or GC–MS.
For this reason, hydrogen appears to be the ideal solution to the GC–MS problem, as we’ve described in an earlier post. But during the panel discussion, the audience expressed a number of concerns about using hydrogen, and the figure below summarises the relative proportions of questions raised:
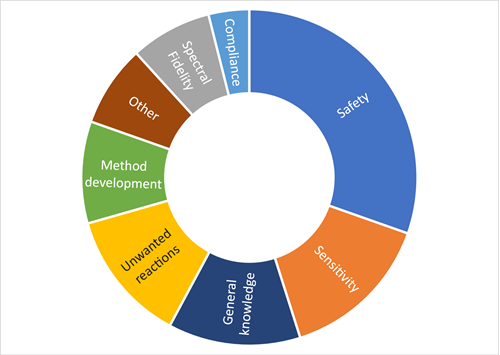
To dig into this in a bit more depth, I’ve picked out some of the most interesting questions received before and during the discussion, and summarised the responses from the panel, which included:
- Diane Turner – Anthias Consulting
- Chris Siegler – Dow, Inc.
- Helen Martin – Markes International
- Matthew Klee – XO Associates (Chair).
You can watch the forum on-demand on the Separation science website.
Some of my colleagues are nervous around safety with hydrogen carrier gas. What is your experience?
The issue of safety accounted for over >30% of questions submitted, and the panel definitely understood the reason for this concern.
Having said that, the panel were very comfortable with using hydrogen as a carrier gas, especially when using modern GCs that have inbuilt features for hydrogen. It was pointed out that a high proportion of GC customers already use GC–FID, which uses hydrogen as a fuel gas, so labs typically have hydrogen plumbed into the lab already. Therefore using hydrogen as a carrier gas does not increase safety concerns greatly, although directing your split to ventilation is recommended.
Also, the panel said that there are lots of safety features developed to support use of hydrogen as a carrier gas, such as:
- Shutdown mode will be activated once instruments detect a leak (which they’re pretty good at doing)
- In-line excess flow limiters can mitigate the risk of leaks between the gas cylinder and the GC
- Hydrogen sensors are available for the GC and the lab
- On-demand hydrogen generators can add another degree of safety, by limiting the volume of hydrogen produced in the event of a leak.
From Markes’ perspective, our Multi-Gas range of TD instruments have been independently certified (by CE and MET) to never reach the lower explosion limit for hydrogen. Having gone through the rigorous process of designing-in this capability and achieving this certification, we’re confident in using hydrogen as a carrier gas.
I’ve heard you lose sensitivity with H2 when using GC–MS. Do you have any tips for minimising this effect?
The panellists explained that hydrogen gives much sharper peaks and allows faster analysis, which results in less longitudinal diffusion of analytes. This in turn means less band broadening, which yields higher S:N ratios and so should keep sensitivity high.
But nevertheless, it is well-known that in some cases you can lose sensitivity when using hydrogen as a carrier gas for GC–MS. So where does the problem lie?
The panellists said that the main cause for loss of sensitivity with hydrogen is actually due to the MS vacuum system. Unfortunately, the optimal GC flow required for good peak shape (>1 mL/min) doesn’t overlap with the optimum MS flow (<1 mL/min) needed to achieve the correct vacuum levels. So analysts need to find a balance between these two conflicting demands – and it was noted that some bench-top TOF MS instruments can cope with 2.5 mL/min without affecting pump efficiency.
If you’re using GC–FID, the panellists said that sensitivity isn’t typically affected by hydrogen carrier gas, but it’s worth reducing your hydrogen fuel gas input to compensate for the carrier flow. The panel said that in lots of incidences they actually experienced an increase in sensitivity when using hydrogen for GC–FID, due to improved peak shape.
Another point was that the loss of sensitivity that can be experienced when using hydrogen for GC–MS is to some degree mitigated by using a preconcentration technique such as TD. Thermal desorption by its nature is so effective at preconcentrating samples that large splits are often required to prevent the detector from becoming saturated. So if any decrease in sensitivity needs to be offset, then this is easily done by reducing the split ratio.
But finally, and on a fundamental level, if an existing helium-based method is more than sensitive enough to cope with your samples, then switching to hydrogen is unlikely to have a major impact on what you can achieve.
I’m concerned over a reduction in data quality with hydrogen. I’ve heard mass spectra can change and there can be unwanted side-reactions – how do you manage these?
Helen Martin from Markes explained that they’ve run a number of applications using hydrogen – including PAHs, PAMS, hydrogen fuels, TO-17 and TO-15 – and not had any problems with side-reactions. TO-15 and TO-17 were a particularly rigorous test, as the standards for these analyses include a mix of halogenated, unsaturated and aromatic hydrocarbons. But in all instances the peak shape and reproducibility were excellent, and there was no loss in sensitivity. Furthermore, she said that, by using sample re-collection on the TD instrument, they were able to confirm that no side-reactions such as hydrogenation or de-chlorination had taken place.
From the perspective of the panel and our own TD–GC–MS studies at Markes, we’ve found the spectral matching to be very good for PAHs, hydrogen fuels, TO-17 and TO-15 when using hydrogen. As discussed by the panel, degradation doesn’t necessarily mean that the analysis has failed – it just needs to be taken account of. One potential option is to change the quantifying and qualifying ions to ones that aren’t affected – as long as spectra, by-products and the selected ions are always produced in the same ratio, then analyses should still be reproducible.
Rounding up
In conclusion, although there are some things to be aware of in terms of safety, sensitivity and side-reactions, it’s important not to overplay these. Hydrogen offers many benefits as a carrier gas for GC–MS and TD–GC–MS, both chromatographically and in workflow terms, such as:
- Sharper peaks
- Higher flow rates
- Faster run times
- Lower cost
- Ease of availability (when using generators)
- Sustainability – an increasingly important consideration
Which in our view, mean the benefits definitely outweigh the disadvantages.
If you need further advice on using hydrogen as a carrier gas, or would like us to help you develop a plan to migrate to Markes’ Multi-Gas technology, then please get in touch.




![Markes TD Brochure Hero3 Corepurple[2]](/media/zmscvqds/markes-td-brochure-hero3-corepurple-2.jpg?crop=0.55128205128205132,0,0,0&cropmode=percentage&width=350&height=260&rnd=132399686846500000)





